Iso14971 Risk Management Template : Calameo 2018mar12 Draft Gap Assessment Report Uk000192 002

Risk management has been conducted following the principles laid out in iso 14971, yet since the advent of the new version of en iso Identify the medical device and the scope of the risk study. For the ce marking of the medical devices, risk management is an essential requirement as per the eu medical device regulation. While convenient from a tool perspective, this approach does not align with iso 14971. This template will provide you with a framework to complete your risk management plan. Iso 14971 risk management process has 10 main clauses and 3 annexes. Annex f of the iso 14971:2007 standard provides a template that may be used for a risk management plan, and the iso/tr 24971:2020 guidance is expected to provide similar content once it is released. Medical device and fda regulations and standards news: In general, the aim is to identify hazards, assess and evaluate the associated risks, control these risks and monitor the effectiveness of risk management measures.
Medical device and fda regulations and standards news: Risk management has been conducted following the principles laid out in iso 14971, yet since the advent of the new version of en iso Risk management is an important lifecycle product development requirement for all medical device organizations when developing, manufacturing, and commercially distributing medical products. Reports generated by imsxpress comply with iso 14971 requirements for risk management file (clause 3.5) and provide most of the content required for that file.

According to the new edition of the risk management standard, iso 14971:2019 the following are six steps in risk management.
Planned risk management activities with the identification of the risk acceptability. While convenient from a tool perspective, this approach does not align with iso 14971. Über 7 millionen englischsprachige bücher. It is expected that tr 24971 will become essential for risk management for medical devices and it will contain all the annexes which are not currently present on iso 14971. Information for safety and information about residual risk iso 14971:2019 overview of structure and contents iso 14971:2019 Iso 14971 medical device risk management software is a windows application for implementing risk analysis, risk evaluation, and risk control in strict compliance with the iso 14971:2012 standard. Schnelle erstellung von geschäftsdokumenten mit dynamischen vorlagen von templafy The risk analysis method shall be described in the risk management plan. Reports generated by iso software comply with iso 14971 requirements for risk management file (clause 3.5) and provide most of the content required. The process flow for risk management based on iso 14971 is shown in figure 1. This includes software as a medical device and in vitro diagnostic medical devices. Iso 14971 risk management for medical devices: Learn how to work with risk management according to the iso 14971:2019 standard. Annex f of the iso 14971:2007 standard provides a template that may be used for a risk management plan, and the iso/tr 24971:2020 guidance is expected to provide similar content once it is released.
Risk study is developed in seven steps: Risk management process, and fundamental risk concepts. Annex f of the iso 14971:2007 standard provides a template that may be used for a risk management plan, and the iso/tr 24971:2020 guidance is expected to provide similar content once it is released. Über 7 millionen englischsprachige bücher.

According to clause 3 in iso 14971, top management must:
Medical device and fda regulations and standards news: Risk management is a fundamental step for medical device manufacturers to demonstrate compliance with the eu directives for medical devices, ensuring the safety of patients and users. Iso 14971 and risk management. The risk analysis method shall be described in the risk management plan. The process flow for risk management based on iso 14971 is shown in figure 1. A risk management report summarizing the results of risk management activities; According to the new edition of the risk management standard, iso 14971:2019 the following are six steps in risk management. Über 7 millionen englischsprachige bücher. Learn how to work with risk management according to the iso 14971:2019 standard. It is expected that tr 24971 will become essential for risk management for medical devices and it will contain all the annexes which are not currently present on iso 14971. Two approaches to integrating fmea with risk management. Establish a list of generic hazards that may apply to the medical device. Risk management plan template (medical device and iso 14971) february 1, 2021 by mathilde émond 24 posts related to risk management plan template (medical device and iso 14971) Annex f of the iso 14971:2007 standard provides a template that may be used for a risk management plan, and the iso/tr 24971:2020 guidance is expected to provide similar content once it is released. Iso 14971 risk management process has 10 main clauses and 3 annexes.
Reports generated by imsxpress comply with iso 14971 requirements for risk management file (clause 3.5) and provide most of the content required for that file. The focus of this blog post is the first of these six steps: According to clause 3 in iso 14971, top management must: Iso 14971 is the key to effective risk management for medical devices. Risk management is a fundamental step for medical device manufacturers to demonstrate compliance with the eu directives for medical devices, ensuring the safety of patients and users. The template includes topics as required by clause 3.4 of iso 14971:2007 and en iso 14971:2012. Risk management plan template introduction document overview references project references standard and regulatory references risk management during software development organization and responsibilities qualification of personnel objective of risk management. Planned risk management activities with the identification of the risk acceptability. Iso 14971 risk management for medical devices: While convenient from a tool perspective, this approach does not align with iso 14971.
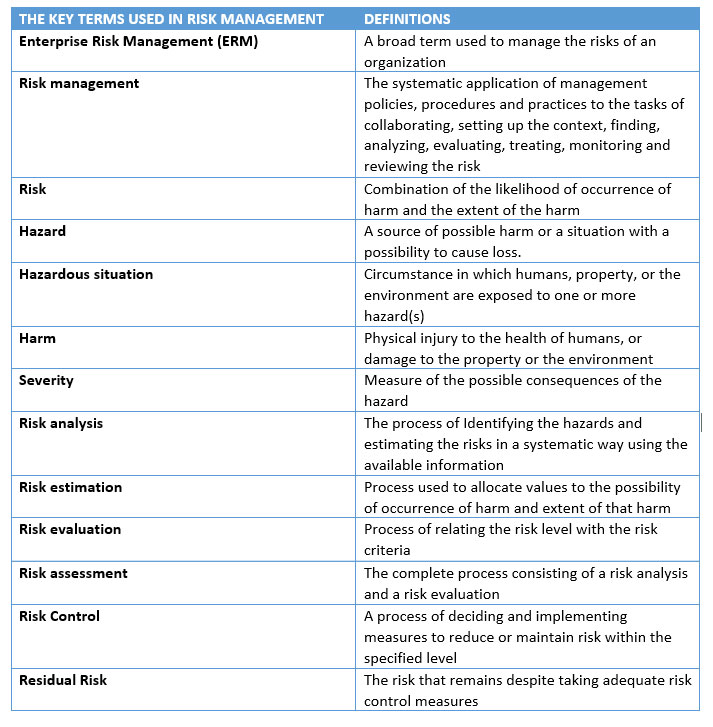
To effectively meet regulatory requirements, manufacturers must utilize the harmonized standard, en iso 14971:2019 risk management standard and the technical report that accompanies it, iso tr 24971:2020.
The iso 14971 is the standard for the application of risk management for medical devices. Iso 14971 risk management for medical devices: This template will be compliant with iso 14971 requirements if you: Guidance on risk analysis process for biological hazards j. The internationally accepted standard guideline for medical device risk management is the iso 14971 standard. Reports generated by iso software comply with iso 14971 requirements for risk management file (clause 3.5) and provide most of the content required. It may also be used as a benchmark on your existing plan. Do not hesitate to download the product! Iso 14971 is the risk management standard for medical devices. It is expected that tr 24971 will become essential for risk management for medical devices and it will contain all the annexes which are not currently present on iso 14971. For the ce marking of the medical devices, risk management is an essential requirement as per the eu medical device regulation. The plan provides a roadmap for the risk management activities to be conducted during the life cycle of the medical device. Risk management process, and fundamental risk concepts. Iso 13485 defines risk based on iso 14971 as the combination of the probability of occurrence of harm and the severity of that harm. risk management process through iso 14971. To effectively meet regulatory requirements, manufacturers must utilize the harmonized standard, en iso 14971:2019 risk management standard and the technical report that accompanies it, iso tr 24971:2020.

Über 7 millionen englischsprachige bücher.

This template will be compliant with iso 14971 requirements if you:
Iso 14971 is the risk management standard for medical devices.

This process intends to include the following steps:
Information for safety and information about residual risk iso 14971:2019 overview of structure and contents iso 14971:2019

Results of risk analysis generated by isoxpress iso 14971 risk management software.

Annex f of the iso 14971:2007 standard provides a template that may be used for a risk management plan, and the iso/tr 24971:2020 guidance is expected to provide similar content once it is released.

All risk management activities must be planned.

Iso 14971 is the risk management standard for medical devices.

To effectively meet regulatory requirements, manufacturers must utilize the harmonized standard, en iso 14971:2019 risk management standard and the technical report that accompanies it, iso tr 24971:2020.

Über 7 millionen englischsprachige bücher.

Risk management is a fundamental step for medical device manufacturers to demonstrate compliance with the eu directives for medical devices, ensuring the safety of patients and users.

It is important to recognize that

This includes software as a medical device and in vitro diagnostic medical devices.
It also includes topics that should be addressed for software risk management according to iec 62304:2006.

Risk management is a fundamental step for medical device manufacturers to demonstrate compliance with the eu directives for medical devices, ensuring the safety of patients and users.
Reports generated by iso software comply with iso 14971 requirements for risk management file (clause 3.5) and provide most of the content required.

Do not hesitate to download the product!

One of the key activities related to risk management is the risk analysis.

It is expected that tr 24971 will become essential for risk management for medical devices and it will contain all the annexes which are not currently present on iso 14971.

The iso 14971 standard was developed specifically for manufacturers of medical devices on the basis of established risk management principles developed over many years.

Iso 14971 medical device risk management software is a windows application for implementing risk analysis, risk evaluation, and risk control in strict compliance with the iso 14971:2012 standard.

Here at qualitymeddev we have a risk management plan template fully editable in word that can be used as starting point for the construction of your risk management process, fully compliant with iso 13485, fda regulations 21 cfr 820, iso 14971:2019 and aligned with the eu mdr 2017/745.

Where does fmea fit in your iso 14971 risk management process?

Learn how to work with risk management according to the iso 14971:2019 standard.

Guidance on risk management for in vitro diagnostic medical devices i.

Risk management is an important lifecycle product development requirement for all medical device organizations when developing, manufacturing, and commercially distributing medical products.

Iso14971 risk management template / fmea compared with risk management according to iso 14971 :

One of the key activities related to risk management is the risk analysis.
Iso 14971 and risk management.

Learn how to work with risk management according to the iso 14971:2019 standard.

Establish a list of generic hazards that may apply to the medical device.

It describes a risk management process to ensure that the risks are known and dominated by medical and are acceptable when compared to benefits.
Iso 14971 risk management process has 10 main clauses and 3 annexes.

Medical device and fda regulations and standards news:
Posting Komentar untuk "Iso14971 Risk Management Template : Calameo 2018mar12 Draft Gap Assessment Report Uk000192 002"